
Optimal thermal cycles for internal combustion engines
The study of the ideal cycle (ideal gas) gives a picture of the efficiency of the cycle used in internal combustion engines. The cycle is considered ideal if the following conditions are met:
– There is no gas loss, and there is no gas left
– An ideal gas with stable thermal capacity coefficients and the ratio between them is 1.4
– Quick addition and loss of heat
– There is no loss in flow
* Internal combustion engines do not work according to Ideal cycles use real gases as they are subject to flow losses, heat losses, and mechanical losses.
Thermal cycles:
The internal combustion engine operates according to one of the following cycles:
- Otto cycle (proven size) your Constant Volume Cycle ( from Otto Cycle)
- Diesel cycle (proven pressure) your Constant Pressure Cycle (Diesel Cycle)
- Dual Cycle Duel combustion cycle
- Atkinson cycle Atkinson cycle
The thermal cycle consists of several thermal procedures taking place on the system (gas), so that the system (gas) returns to the same thermodynamic state with which it started.
Thermal procedures:
- Conducting constant temperature Isothermal process (constant temperature)
- Conducting proven Size Constant Volume process
- Conducting evidence of pressure Isobaric process (constant pressure)
- The non – heat transfer (Odipati) Adiabatic process the (First Heat Transfer)
| Temperature verification procedure | |
 |
In this procedure there is no change in temperature and when the first law of thermodynamics is applied
| (1) |
In the absence of a change in temperature, no change occurs in the internal energy of the gas, and the equation becomes as follows:
| (2) |
The amount of heat transferred equal to the work is equal to:
| (3) |
The relationship between pressure and volume at different points in the procedure is as follows:
| (4) |
where:
D U = change in the internal energy of the gas
Q = amount of heat transferred
W = work
V = volume
P = pressure
P 1 = pressure at state 1, V 1 = volume at state 1
P 2 = pressure at state 2, V 2 = size at case 2
| – procedure proven Size | |
| In this procedure, the volume does not change, so the gas does not do any work. When applying the first law of thermodynamics (1), and in the absence of work, W is zero. |  |
The equation becomes:
| (5) |
The amount of heat transferred to and from the gas is:
| (6) |
The procedure for verifying the size follows the path:
| (7) |
The relationship between pressure and volume at different points in the procedure is as follows:
| (8) |
Where:
c V = specific heat capacity with constant volume
P 1 = pressure at state 1, T 1 = temperature at state 1
P 2 = pressure at state 2, T 2 = temperature at state 2
n = number of moles in the gas
D T = The temperature difference
during this procedure does not move the piston, whether at the lower dead point or the upper dead point.
During this procedure the gas pressure remains constant and the procedure follows the following path:
| (9) |
The relationship between pressure and volume at different points in the procedure is as follows:
| (10) |
The amount of work done is
| (11) |
where:
D V = change in volume
and V 2 , V 1 amount of volume at points 1, 2 in order
and T 2 , T 1 amount of temperature at points 1, 2 in order
The equation becomes:
| (12) |
The adiabatic procedure follows the path:
| (13) |
The relationship between pressure and volume, temperature and volume, and pressure and temperature at different points in the procedure is as follows:
| (14) | ||
| (15) | ||
| (16) |
and p 2 , p 1 the amount of pressure at points 1, 2 in order
and V 2 , V 1 the amount of volume at points 1, 2 in order
and T 2 , T 1 the amount of temperature at points 1, 2 in order
g The ratio between the two heat capacities (relative ) gas
during this procedure piston moves from the lower dead point to the upper dead point. From a volume equal to the total volume V t , to the volume of clearance V C .
And called the ratio between the total size of the cylinder V ‘s t (plus size cylinder size clearance) to the size of the clearance VC Compression ratio r .
| (17) |
Ideal air cycles
In these cycles, we assume that air is used instead of the air-fuel mixture and take into account the following:
– There is no heat transfer between the surfaces of the engine and the air
– The combustion is instantaneous and complete
– The specific heat capacity is considered constant.
Volume Stabilization Cycle (Otto)
| Spark ignition engines follow the Otto cycle but on real gases (a mixture of air and fuel) rather than air. The Otto cycle consists of four thermal procedures (adiabatic – constant volume – adiabatic – constant volume) | |
| Depate procedure (no heat exchange occurs) from point 1 to point 2(charge compression, work done on gas) | 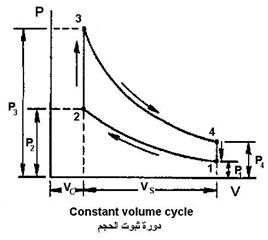 |
The amount of work done on the gas is W in .
| (18) |
2. The volume constancy procedure from point 2 to point 3
(adding heat at constant volume, combustion)
and the amount of heat added to the gas is Q in
| ( 19 ) |
3. An adiabatic procedure (no heat exchange occurs) from point 3 to point 4
(charge expansion, work done by the system)
and the amount of work done by the gas is W out
| (20) |
4. Volume stabilization procedure from point 4 to point 1
(heat disposal at constant volume, exhaust)
and the amount of heat withdrawn ( disposal ) from the gas is Q out
| (21) |
* Therefore, the total work done on the cycle W is equal to the work done by the gas minus the work done on the gas (the area confined inside the cycle)
| (22) |
| (23) |
Where:
g = the ratio between the two heat capacities (constant pressure and constant volume)
and p 4 , p 3 , p 2 , p 1 the amount of pressure at points 1, 2, 3, 4 respectively
and V 4 , V 3 , V 2 , V 1 The amount of volume at points 1, 2, 3, 4 in the order
c v Specific heat capacity of the gas at constant volume
* The efficiency of the cycle h is equal to the work of the cycle divided by the given heat, where the work is equal to the difference between the given heat and the heat lost (second law of thermodynamics ):
| (24) | ||
| (25) | ||
| (26) |
which is equal to h air Standard Efficiency (ASE)
| (27) |
where T 4 , T 3 , T 2 , T 1 is the amount of temperature at points 1, 2, 3, 4 in order.
Practical example
using Excel for a constant-volume air cycle engine (Otto)
Input information:
* Initial state of the gas (point 1):
t_1 = Initial temperature [°C] – the program will convert it to Kelvin
p_1 = Initial absolute pressure of the gas [kPa (kN/m)]
* Engine Specification (Point 1):
V_1 = Gross Cylinder Volume [cc]
* Engine Specification:
rr = Compression Ratio
* Operating Condition (Point 3):
t_3 = temperature [degrees Celsius] – where the program will convert it to Kelvin
* gas specifications
g = ratio between the two heat capacities (constant pressure and constant volume)
* practical Excel program for the Otto cycle
Stabilized pressure cycle (diesel)
| Compression ignition engines follow the diesel cycle but on real gases instead of air. The diesel cycle consists of four thermal procedures (adiabatic – constant pressure – adiabatic – constant volume) | |
| An adiabatic procedure (no heat exchange occurs) from point 1 to point 2The amount of work done on the gas is as in equation (18) (charge compression, work done on the systemPressure stabilization procedure from point 2 to point 3(Adding heat when pressure is stable, combustion) | 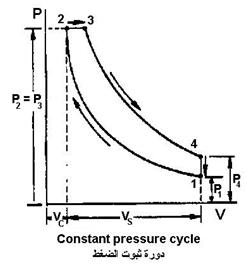 |
The amount of heat added to the gas is Q in .
| (28) |
The amount of work done by the gas during this procedure is W out (1)
| (29) |
- An adiabatic procedure (no heat exchange occurs) from point 3 to point 4
(charge expansion, work done by the system)
The amount of work done by the gas is as in equation (20)
- Measure of constancy of volume from point 4 to point 1
(heat disposal when volume is fixed, exhaust stroke)
and the amount of heat withdrawn (disposal ) from the gas as in equation (21)
* Therefore, the total work done for the cycle W is equal to the work done by the gas minus the work done on the gas as in equation (22 ) and equal to
| (30) |
And the efficiency for the session is as in Equation (24)
| (31) |
Where:
c p Specific heat capacity of the gas at constant pressure
Practical example
using the Excel program for a diesel engine running at a constant pressure air cycle (diesel)
Entered information:
* Initial state of the gas (point 1):
t_1 = Initial temperature [°C] – where it will The program converts it to Kelvin
p_1 = the absolute initial pressure of the gas [kPa (kN/m²)]
* engine specifications (point 1):
V_1 = gross cylinder volume [cc]
* engine specifications:
rr = compression ratio
* operating condition (Point 3):
t_3 = Temperature [°C] – the program will convert it to Kelvin
g = Ratio between the two heat capacities (constant pressure and constant volume)
* Practical Excel program for a diesel cycle
Dual cycle
| High-speed, full-load compression ignition engines burn a fraction of the fuel at constant volume and a fraction at constant pressure. The cycle is similar in this case to the double cycle. | |
| The dual cycle consists of five thermal procedures 1. An adiabatic procedure (no heat exchange occurs) from point 1 to point 2 (charge compression, work done on the gas) and the amount of work done on the gas is as in equation (18) 2. Volume constant ( No work occurs) from point 2 to point 3 (adding heat when volume is constant) and the amount of heat added is as in equation (19) 3. The procedure for constant pressure from point 3 to point 4 (adding heat at constant pressure, combustion) and the amount of heat The added gas is Q in as in equation (28) and the amount of work done by the gas is as in equation (29) 4. An adiabatic procedure (no heat exchange occurs) from point 4 to point5 (charge expansion, work done by the system) and the amount of work done by the gas is as in equation (20) 5. The volume constant procedure from point 5 to point 1 (heat removal at constant volume, exhaust stroke) and the amount of heat withdrawn as in equation (21) | 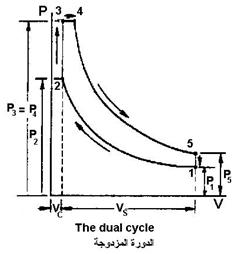 |
* and the total turnover is W
| (32) |
And the efficiency for the session is as in Equation (24)
| (33) | ||
| (34) |
Practical example
using Excel software for a dual- air cycle engine
Input information:
* Initial state of the gas (point 1):
t_1 = Initial temperature [°C] – where the program will convert it to Kelvin
p_1 = The absolute initial pressure of the gas [kPa (kPa) kN/m²)]
* engine specifications (point 1):
V_1 = gross cylinder volume [cc]
* engine specifications:
rr = compression ratio
* operating condition (point 3):
t_3 = temperature [°C] – where The program will convert it to Kelvin,
g = the ratio between the two heat capacities (constant pressure and constant volume)
* Practical Excel Double Course
Atkinson cycle
| This cycle is similar to the Otto cycle in that heat is added at constant volume, but differs from it in that the ratio of compression (the volume at point 2 divided by the volume at point 3) and the rate of expansion (the volume at point 5 divided by the volume at point 4) are not equal . This is done by opening the intake valve for a longer period of time, and this leads to an increase in the rate of expansion, allowing to benefit from most of the energy in the gas during this procedure. Usually, the compression ratio is in the range of 10 and the expansion ratio is in the range of 20. The Atkinson cycle gives efficiency at the expense of power, and gives fuel savings. The presence of return gas from the cylinder in the intake manifold leads to its entry into the next cylinder in the order of the fire, which reduces the energy loss resulting from the throttling. | |
| The Atkinson cycle consists of five thermal proceduresThe procedure for moving the piston forward and keeping the intake valve open for a longer period of time 1-2 (no work occurs as the cylinder has not yet been closed)2. An adiabatic action (no heat exchange occurs) from point 2 to point 3 (charge compression, work done on the gas) |  |
The amount of work done on the gas is W in .
| (35) |
3. Consistent volume procedure (no work occurs) from point 3 to point 4
(adding heat when volume is constant)
and the amount of heat added is as in equation (19) with point 2 replaced by 3 and 3 by 4
4. Adiabatic procedure (does not occur Heat exchange) from point 4 to point 5
(charge expansion, work done by the system)
and the amount of work done by the gas is W out as in equation (20) with point 3 replaced by 4 and 4 by 5
5. Volume constant procedure from point 5 To point 1
(disposal of heat when volume is fixed, exhaust stroke)
and the amount of heat withdrawn (disposal ) as in Equation (21) with the replacement of point 4 by 5
* and the total work cycle W
| (36) |
Where:
V 2 = cylinder capacity when the intake valve is closed
* and the cycle efficiency is h as in equation (24)
| (37) |
A practical example
using the Excel program for an engine that works according to the air cycle Atkinson
The entered information : (In this program I replaced points 1,2,3,4,5 with the following points 1,1a,2,3,4)
* The initial state of the gas (point 1):
t_1 = Initial temperature [°C] – where the program will convert it to Kelvin
p_1 = Initial absolute pressure of the gas [kPa (kN/m²)]
* Engine specifications (point 1):
V_1 = Gross cylinder volume [cubic centimeters] ]
* Engine specifications:
rr = compression
ratio = ratio between the volume of the cylinder when the intake valve starts to close to the volume of the cylinder
* Operating condition (point 3):
t_ 3 = temperature [°C] – where the program will convert it to Kelvin
g = The ratio between the two heat capacities (steady pressure and constant volume)
* Practical Excel program for the Atkinson cycle
There are no reviews yet. Be the first one to write one.



